Stop wasting time and jeopardizing your production processes with manual, inefficient paperwork. It’s time for you to take back control of your data with an electronic batch record (EBR) designed to easily track chain of custody and chain of identity for all your units.
“We embarked on a comprehensive evaluation of commercial quality system software and after much consideration we decided upon Title21...”
Diane Kadidlo
Executive Molecular and Cellular Therapeutics Facility Director
University of Minnesota
Repurpose your Paperwork
Title21 streamlines your documentation and quality processes so you can focus on delivering more life-saving treatments not more paperwork.
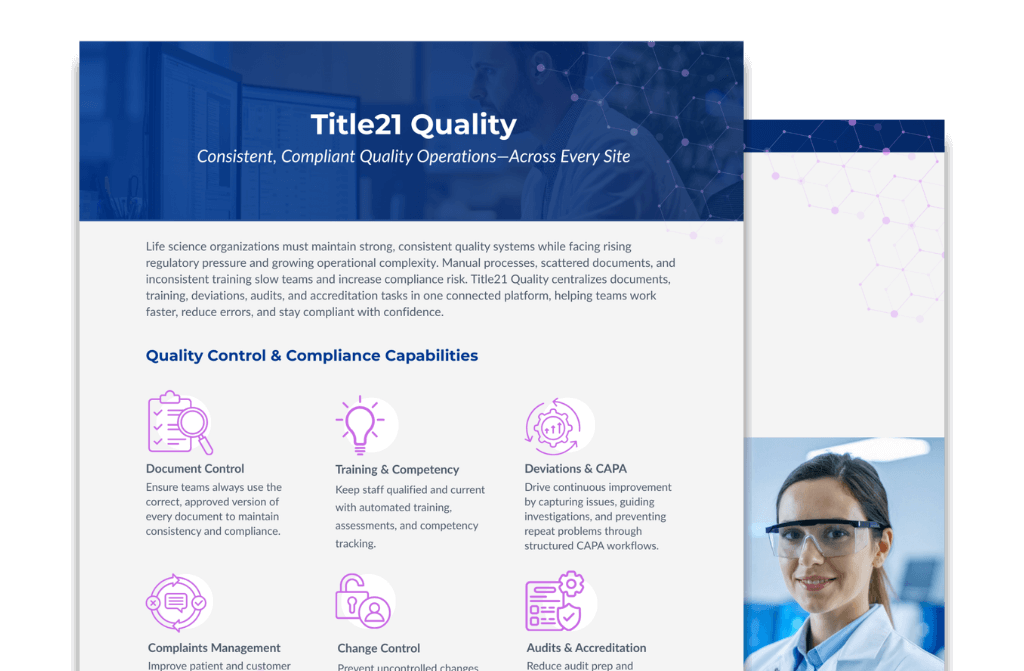
Solution Overview
Enterprise Quality Management System
Streamline your quality processes, minimize administrative burden, and reduce operational risk with a secure, intuitive, and compliant platform for end-to-end quality management. Download your free Solutions Overview today.
Complete the form to download.
Ready to Focus More on Patients and Less on Paperwork?
Learn more on how Title21's industry leading biotherapies software solutions can help your facility improve quality and reduce risk.